What are computer models of disease?
Computer models allow us to simulate complicated behavior on a computer so that we can understand it better. When a working system such as the brain is formed by many (billions!) of interacting elements, things get complicated pretty fast. So much so that, even knowing exactly how each individual element works, we cannot predict what the system as a whole will do. Will it show healthy activity, or enter into epileptic states? With computer models we can simplify the system and simulate its behavior in order to try to answer such questions.
So how do we use computer models in epilepsy research?
Currently, there are several different modelling avenues being explored in epilepsy research. Different models vary in the amount of detail that is put into them, ranging from simple models that indicate whether a region is active or inactive, to models that try to mimic experimentally recorded data in great detail. Our approach has been that taking into account the network perspective is more important that an exact description of what happens locally. Much is still unknown about the particular mechanisms leading to the onset of epileptiform activity and the propagation of this activity. From a clinical perspective, our main focus is the question: how can we prevent the propagation of epileptiform activity?
To answer this question, we first need to simulate how epileptic activity propagates over the brain. This can be done from the network description of the brain: each brain region is represented as an element or node of a network, and the connections or links between them can be estimated from different scans (such as MRI, MEG or EEG). Then, each node is assigned one (or more) dynamical variables. These are governed by mathematical equations describing how each region behaves independently and how it responds to interactions with other regions. Overal, this can give us a view of the local (at each region) and global behavior of the brain. And, most importantly, how it will respond to different treatments.
Then, if the computer model can replicate how brain activity is affected by different interventions, we can use the model to test as many resection strategies as needed. This can be done with the so-called Virtual Resection method, whereby a brain resection is simulated in the model by removing a given set of nodes or links. This will affect the behavior of the system, for instance a given link can act as the main bridge between two regions, and removing it can stop the spreading of epileptiform activity, as it is shown in this figure:
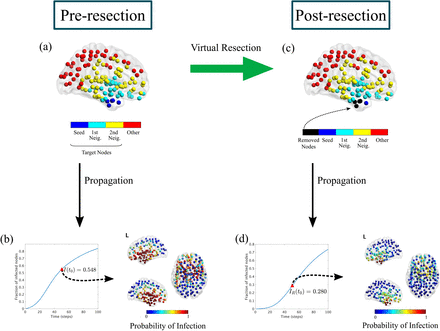
Virtual Resection Implementation. The target nodes for the virtual resection VR are all nodes at two steps or less from the seed, i.e. the seed and its first and second neighbours (panel a). The initial seizure propagation is the fraction of infected nodes at t0 = 50, I(t0) = 0.548 (panel b). A virtual resection of 5 nodes is implemented in the network by setting to 0 all the links with the corresponding nodes, marked in black in panel c. Seizure propagation is now reduced by approximately a factor 2 (IR(t0) = 0.280), and the probability that the seizure reaches regions outside the seed decreases considerably (panel d). This example corresponds to patient 4. Figure copied with permission from Millan et al. 2021.
This method can help us, for instance, in determining whether a larger resection may be beneficial or whether a smaller resection may be enough for a specific patient. Eventually, we expect that the models will also help us finding alternative — personalized — resections for patients for whom standard resections might be inefficient.
What are the difficulties that these computer simulations are facing? First of all, the brain contains billions of neurons, what is the right scale of observation? That is, how large or small should our brain network be? Then, one needs to decide how to construct said network: activity is thought to propagate over the structural network of physical connections, but obtaining large-scale and precise structural networks for each person is difficult, expensive, and slow. What is the appropriate dynamical model for activity in each individual brain region? And how should the resections be implemented in the model? All these questions are currently being explored world-wide in different research efforts.
Do you want to test it yourself? You can test a simple version of our model!